Category Archives: FAQs
The introduction of the da Vinci robot-assisted surgery for use in gynecologic surgery in the past few years has been of great benefit to many patients. It allows the use of a minimally invasive laparoscopic route to cases that would otherwise require an abdominal approach. In the field or reproductive surgery, this benefit most frequently pertains to the removal of uterine fibroids. In the past, the majority of fibroids could only be removed through laparotomy, or a large open incision. But with the use of robotic surgery, the vast majority of these procedures can be performed laparoscopically. This offers the patient a huge benefit. What usually required a 2-3 day hospital stay and 4-6 week recovery is now an outpatient procedure, with the patient going home the same day, and able to return to work in 7 days. There is significantly less blood loss, and less scar tissue formation around the uterus. Surgical outcomes are equal to those performed through an open procedure. With such great benefit, I strive to not perform on open procedure on patients. Even in very difficult settings, nearly all cases can be performed laparoscopically. The only cases which I consider laparotomy instead of a minimally invasive surgery is in those cases with large numbers of fibroids (more than 10), though even with large numbers, select cases may be robotic candidates. Fibroid size is not a limitation.
Another condition that can benefit from the use of robotic assistance is that of severe endometriosis. Particularly in the setting of pelvic pain, it is essential to remove all of the endometriosis. This can be done by standard laparoscopy, but if other organs such as intestines are involved, most surgeons under-treat the disease due to fear of injuring surrounding critical organs. Few surgeons treat it adequately due to the extended amount of time that such a surgery can take. The use of the da Vinci robotic system allows me to more thoroughly treat the disease, and perform a much more careful dissection around critical organs, and do all of this much faster than can be done by standard laparoscopy.
For women facing a hysterectomy, the use of robotic assistance allows for the performance of much more surgically challenging cases. These involve large fibroids, fibroids in difficult locations, endometriosis, and scar tissue. Well more than 90% of abdominal hysterectomies could and should be performed through a minimally invasive route.
You may ask, if this is of such benefit, why isn’t the procedure being done more often? There are several answers to this question. Many times, with general gynecologists, the patients aren’t truly given informed choices of their options. They are usually given the options that their physician can perform, but less often are they given other viable options. Many gynecologists lack the skills and/or training necessary to perform advanced laparoscopic surgery. Unfortunately, they often fail to consider what is in the best interest of the patient, as that may mean referring that patient to an advanced laparoscopic surgeon. Often, the patient may not know all of her options, or be told by the physician that it is not an option. These are situations in which doing your homework or seeking a second opinion can benefit you.
Is the robot necessary for these more difficult cases? No, as most of these above mentioned surgeries can be accomplished by a skilled laparoscopic surgeon. But with increasing complexity, comes the need for more advanced surgical skills, and longer operative times. To a skilled surgeon, the robot does expand what can be “easily” be done laparoscopically, which allows for a faster surgery, improving patient recovery.
If you might be facing a difficult surgery, and question whether the use of robotic assistance may benefit you, please call to arrange a meeting or phone consultation with Dr. Duffy
We do not operate a sperm bank for the general public. Sperm storage is performed at the FIRM for many reasons such as fertility preservation prior to cancer therapy, or for storage to allow couples to continue attempting conception when the male is not physically available. For patients who are looking to purchase sperm for use, we recommend contacting one of the commercial cryobanks (sperm banks). Companies we frequently use are California Cryobank, Fairfax Cryobank, and Xytex. By going to their websites, patients can create an account and select the donor profile that they prefer. When the patient is ready to conceive, that sperm is then shipped to the FIRM where it is subsequently stored until use
We currently do not perform PGD (preimplantation genetic diagnosis/screening) for the sole reason of gender selection. In those patients who are undergoing PGD for other medical indicated reasons, then sex is sometimes also determined at the same time. Some diseases are transmitted through only one sex, thus in those cases, gender selection can be applied.
The following are some of the questions that we received from the audience at our recent Fertility Seminar. Some of these were addressed at that time, but we could not answer all questions. If there are other general questions that you would like to have addressed, please feel free to comment below, and I will answer them. — Dr. Duffy
1. Can you do an HSG (hysterosalpingogram) when a person has an allergy to IVP dye?
Yes, an HSG can still be done. Though the dye usually used is an iodine based dye, the procedure can also be done by using a type of dye normally used for MRIs that contain Gadolinium instead of Iodine.
2. With IVF with ICSI, do you choose any sperm to inject or do you get the best looking one before injecting into the egg?
During the process of ICSI, the embryologist starts by choosing a sperm that is very motile (moving). Most sperm that are significantly deformed aren’t as active. A needle is then used to cut or break the tail of the sperm so that it can be carefully loaded into the needle. This allows a closer look at the sperm as well once the movement stops. Using these techniques, allows us to achieve a very high fertilization rate.
3. In the state of Florida, does a gestational carrier have any legal rights to keep the baby?
One important aspects of third party reproduction, such as using a donor, surrogate, or gestational carrier is having a valid legal contract in place that has been created by lawyers familiar with the process. This routinely addresses the issue of later legal rights, and is clearly laid out in the contract. However, if this part were to be omitted, the carrier could have some legal bearing.
4. How many appointments can we expect for IVF from start to finish?
Once the initial consultation with the physician is performed, the next step is to schedule an appointment in which a “SonoHSG” is performed. This is a special ultrasound which allows us to clearly see that there are not any abnormalities in the uterine cavity that could interfere with the process. A “trial transfer” is also performed at the same time, which takes a measurement of the uterine depth, and ensures that a catheter can be easily placed in to the uterus. On the same day, you will have a consultation with one of our IVF nurses who will lay out the entire process with you, give you a schedule of all the steps, and order your medications. Once you are ready to start the ovarian stimulation, this usually involves between 4-5 ultrasounds to monitor your progress. After that, there is the day of the retrieval, the day of the embryo transfer (3 or 5 days later), and then the scheduled pregnancy test (usually 14-16 days after the retrieval). So in all, there would be about 10 visits for the average IVF cycle.
5. How long does it take from start to finish for an IVF cycle?
Most patients will initiate the IVF process by starting on a birth control pill. This is used to limit any interference that your natural cycle could have on the IVF cycle. This lasts for 3 weeks, at which time the stimulation phase begins, which lasts about 14 days until egg retrieval. A pregnancy test is performed approximately 2 week after egg retrieval. Thus, from starting the birth control pill to a pregnancy test takes approximately 7 weeks.
6. How do you test for ovulation and ovarian reserve in a women who has no uterus who would like to use a gestational carrier?
A woman without a uterus still has a hormonal cycle, and ovulates, but determining this timing is slightly more complicated due to the lack of menses which tells us where she is in her cycle. But with ultrasound monitoring and blood work looking at several hormones, we can still get the information needed. Determining ovarian reserve does not require the uterus to be present and is usually performed with assessments such as an AMH hormone level, and an Antral Follicle Count (AFC), which is determined by ultrasound.
7. How does PCOS affect fertility?
One of the hallmarks of PCOS is the lack of regular ovulation. This can be resolved through the use of medications (oral pills or injectable hormones). However, even once ovuation is established, patients with PCOS still have slightly lower conception rates and slightly higher miscarriage rates than those women without PCOS. The exact cause of this has not been clearly established, though excess androgens or the insulin resistance may play a role. The use of metformin in women who are trying to conceive has been associated with some improvement in pregnancy rates, and decrease in miscarriage rates, though this still does not result in full normalization of pregnancy rates.
8. What monitoring should I have when on a clomid/femara- IUI cycle?
There are several options for monitoring in this situation. Many times, patients may not be regularly ovualating, and in those cases, using an ultrasound to monitor for follicle development is usually the best choice. If a woman has been confirmed to ovulate, and can reliably detect her LH surge with an ovulation predictor kit, then that can be used to time the insemination. However, ovulation predictor kits can be frustrating to use for many women. If they work, then they can be successfully used, however, about 25% or more of women who are known to be ovulating won’t be able to detect the LH surge.
9. How many eggs/follicles do you like to see with a patient on clomid or femara?
The goal of oral ovulation therapy is obviously to induce ovulation. The number of mature follicles seen may vary among patients but usually is 1-2 for most patients. Studies have looked at pregnancy rates in comparison to the number of follicles developed, and with oral medications, it has been found to not be affected by whether there is 1, 2, or 3 follicles developed.
10. How many IUI cycles should you have before considering IVF?
We always try to use our experience and knowledge to help a patient conceive with the easiest, least invasive, and most economical method possible. Doing IUIs (intrauterine inseminations) is recommended for many different reasons as a first line therapy prior to going to IVF. It is difficult to judge exactly when a patient should go to IVF, but we have to take many factors into consideration, such as patient age, prior history, and infertility issues. In most cases, it is felt that most women who will conceive with a given therapy will likely do so in the first 3-4 months. Once a person approaches this time frame, it is logical to start making future plans and weigh options such as continuing with the current therapy vs progressing to IVF. For women who are older, we often encourage this transition earlier, as pregnancy rates continue to decline with age. Younger women have more flexibility in this aspect. There are many more factors to take into consideration, but in general, we will start to bring up the subject on the 2nd or 3rd cycle in order to have a future plan laid out that would best achieve the goal of having a baby.
11. How often can an HSG be performed, and is it recommended to keep the tubes open for conception?
We don’t recommend repeating an HSG unless something has changed recently, such as a pelvic infection, ectopic pregnancy, or other situations which could have caused scar tissue damage to the tubes. Pregnancy rates have been suggested to be slightly higher following an HSG, but there is not any role for repeating the test.
12. How important is sperm morphology in the setting of a good sperm count?
Criteria for what constitutes a “normal” sperm morphology percentage has changed in recent years, with criteria becoming more strict. Isolated cases of low morphology, with otherwise good counts are less troubling than having multiple abnormalities. Trying inseminations is a first line therapy, which may help in these cases, as a lot of the abnormal sperm are washed out in the preparation process, thus likely increasing the percentage of good sperm. With a good overall sperm number, even if the percentage of normal sperm is low, you will ultimately have a reasonable number of good sperm. To summarize, a small percentage of a big number is still a pretty big number. But if overall sperm numbers are low, and morphology is low, then conception rates may not be as good. But the primary numbers that we look at to judge conception chances for inseminations are still the total motile sperm count, which is calculated from the volume, total count, and percent moving. This has always had the highest predictive ability for predicting the chance of conceiving with inseminations.
13. Are egg donors from the same city?
Because of the close monitoring needed for egg donors, nearly all of our anonymous donors come from the greater Jacksonville area. We choose to limit the number of times that one person can donate to a total of four. There are no specific scientific reasons on limits in this area, but we feel that it is a reasonable limit. Many donors are split between two recipients. It is hard to estimate the number of children from each donor, but that number is likely to be a maximum of 6-8 from one donor who has donated the maximum number of foiur times.
14. How is the use of Testosterone (Testim, Androgel, etc) affecting sperm production?
I was asked this question by several people after the seminar in response to my comments during my male fertility lecture. The number of men that are treated with testosterone replacement continues to rise. The physician prescribing this may be a primary care physician or a urologist. Often they do not ask, or do not know to ask on plans for conception. The use of testosterone in any form (patch, gel, or injection) is associated with a rapid shut down of sperm production. This fact may not be known by all primary care physicians, but is known by all urologists. In most cases, this is reversible, but it can be expected to take 4-6 months to fully recover sperm counts. Men who are being treated with testosterone and wish to conceive should be evaluated by a reproductive physician. There are measures that can be taken to help get sperm production started back, but an endocrine evaluation is often necessary to look into reasons as to what may be affecting the normal testosterone production (the reason the medication was started in the first place).
15. Do you recommend an endometrial biopsy before an IVF cycle to theoretically increase the chance of successful implantation of an embryo?
This is a new area of interest in which I have participated in the past. It has been found by small studies that disrupting the endometrium before or during an IVF cycle may help to improve implantation rates. This injury stimulates the production of many chemicals and repairing mechanisms in the endometrium. There are several ways to cause an injury, but most usually would be by endometrial biopsy. I offer this procedure to my patients who have had otherwise unexplained implantation failures. However, because this procedure is painful, and not tolerated well by many patients, I do not think that it is justified in every IVF cycle. Hopefully future studies will help us determine the best way in which to do this procedure and what patients would benefit the most from it.
The process of IVF involves an investment of time, emotional stress, and expense. When a couple has gone through this process, they would like to ensure that they have the best chance possible of reaching their goal of having a baby. However, the goal of IVF is to help the couple have a single baby, though we recognize that twin pregnancies are not uncommonly the outcome. Ideally, each embryo would produce a live born baby, but unfortunately, this is not the case. Many factors may affect an embryo’s ability to continue to grow normally. Because of this fact, often more than a single embryo is transferred back to the uterus.
The decision on how many embryos to transfer is a difficult one that tries to reach a balance between having a pregnancy and ultimately a live birth, but not having a multiple pregnancy. This decision is a joint plan between both the couple and the physician, and is reached only after a thorough discussion of the many critical aspects involved.
Embryo transfers most commonly occur on the third day (cleavage stage) or fifth day (blastocyst stage) following egg retrieval. Ideally, embryo transfer would always occur at the blastocyst stage, as these embryos have a higher success rate, which can allow for the transfer of fewer embryos, thereby giving a higher pregnancy rate while limiting the number of multiple pregnancies. Unfortunately, not all patients will have enough high quality embryos to make it to the 5th day for transfer. In these patients, we often feel that transferring the embryos on the 3rd day will provide a reasonable pregnancy rate in those situations in which extended culture would not have provided an adequate number of surviving blastocysts.
Factors that should be considered are many, including the age of the woman (or the egg donor), previous reproductive outcome, previous IVF outcome, quality of the embryos, day of transfer, the couple’s reproductive goals, patient body habitus, and any co-existing complicating medical conditions.
The ASRM (American Society for Reproductive Medicine) has published guidelines on the recommended number of embryos to transfer.
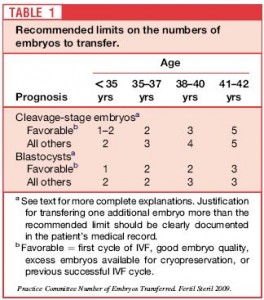 These guidelines have been established based on success data in an effort to reach a reasonable balance between IVF success and limiting multiple gestations.
These guidelines have been established based on success data in an effort to reach a reasonable balance between IVF success and limiting multiple gestations.
Though not legally required, it is strongly suggested that deviations from these numbers only occur in certain circumstances.
More recent data is being published demonstrating the benefits of transferring only a single good quality blastocyst. This should be considered in the under 35 group with a favorable outcome, and more especially for those under 30. Single embryo transfer can give very satisfactory pregnancy rates, while maintaining very low rates of twins (<3%). This is compared to the twin rate by transferring two blastocysts, which can be 25-30%, with a ~4% rate of triplets (if one embryo should split on its own).
In summary, the decision on the number of embryos to transfer must be made between the patients and physician taking into account many different factors, and should be undertaken after thorough consideration of the risks involved.
Many patients have heard of IVF (In vitro fertilization), and may even be facing IVF in their effort to conceive. However, many do not know exactly what is involved. This will hopefully give a quick overview of a typical IVF cycle.
General
The basic idea in IVF is to give the patient a large amount of gonadotropins (the hormones from the brain that stimulate egg development) to cause the development of many follicles (the ovarian cysts that each contain an egg). The hormones used must all be given as injections, as they do not come in pill form. The injections are given just under the skin like an insulin injection that a diabetic person might take. Once the follicles are fully developed, which usually takes 10-12 days of medication, the eggs are retrieved through an in-office procedure. The eggs are given to the embryology lab where they are fertilized with sperm, and allowed to grow for 3-5 days. At that point, the embryo(s) are transferred back into her uterus through a small catheter (tube).
Stimulation Protocols
There are several basic plans (protocols) for performing ovarian stimulation for IVF. There have been numerous variations reported over the years, but primarily two or three basic standard types have proven to be the most successful. The differences between any of the protocols are subtle. It is usually only through the judgment of the physician that it can be determined which protocol will be most suitable for a particular patient.
The two protocols that have been time-tested and most often used are termed “Agonist Protocols” and “Antagonist Protocols”. These terms refer to the type of medication that serves to prevent your body from trying to prematurely ovulate or release the developing eggs. In both protocols, the stimulation medication (FSH and LH) are used for a total of 10-12 days.
Though it may sound counterproductive, it is helpful to use a birth control pill for at least one cycle before starting the IVF medications. This serves to keep the ovaries quiet and prevent any cyst formation that may later interfere with the IVF cycle.
In an Agonist Protocol, a hormone (called a GnRH-agonist) is used to prevent the brain from trying to prematurely ovulating the developing follicles. This is usually started during the last week of the birth control pills, and continued daily up until the point of egg retrieval. The stimulation medications (FSH and LH) are started after being on the GnRH Agonist for approximately one week.
In an Antagonist Protocol, the stimulation medication is started once the menses following the birth control pill cycle occurs. After 4-6 days of stimulation medication, another hormone is started (a GnRH-antagonist), which prevents premature ovulation of the developing follicles. This is also continued until the point of egg retrieval.
Cycle Monitoring
The initial dose of medication is chosen by the physician based on several factors, including the patient’s age, previous IVF experience, and hormone levels. Usually the dosages of stimulation medications will be adjusted during the stimulation phase based on the results of close monitoring which includes a pelvic ultrasound and hormone measurements. This monitoring is performed at baseline before starting stimulation, and after the first 3-5 days of stimulation medications. From that point on, an ultrasound and estrogen blood level is usually performed every 2-3 days until the time of retrieval, for a total of 4-5 monitoring visits.
Once the majority of the ovarian follicles reach the proper size (usually 17-19 mm in diameter), a separate medication is given (hCG- human chorionic gonadotropin). This serves to “trigger” the eggs, causing the eggs to genetically awaken from their idle state, continue development, and become “mature” or able to be fertilized and develop into an embryo.
Egg Retrieval
The hCG medication is given with specific timing. If the eggs are not retrieved, they would be ovulated and released. This timing is such that the egg retrieval occurs just several hours before the eggs would be released. The egg retrieval occurs in our certified operating room located within our office. Anesthesia medication is administered through an IV placed in an arm vein. This allows the patient to be fully relaxed and comfortable during the procedure. A pelvic ultrasound is performed as usual, and ultrasound guidance is used to guide a needle into each ovary allowing the fluid (containing the egg) in every follicle to be removed. After the eggs are carefully removed, they are given to the embryologist (the laboratory scientist specially trained in handling eggs and embryos). The entire retrieval usually takes about 15-30 minutes, and the patient goes home once fully recovered from the anesthesia (about 45 minutes later).
Fertilization and Embryo Culture
Once the eggs are retrieved, they are fertilized several hours later. This is usually done by mixing the eggs with the sperm. In cases of male fertility issues, an individual sperm may be directly injected into each egg. The fertilized eggs then start the process of dividing. They are grown (cultured) in a very closely monitored and controlled setting which allows for the optimal environment for the early embryos. They are assessed on the second day after retrieval, and depending on several factors, a decision will be made by the physician to either transfer the embryos on the following day (Day 3), or allow them to grow for an additional two days and transfer them on Day 5.
Embryo Transfer
When the day comes for the embryo transfer, you will return to the office. The transfer will take place in another room specifically used for this purpose which is located next to the embryology lab. The transfer is a simple and painless procedure that does not require any type of anesthesia. It is very similar to having a pap smear performed. During the transfer procedure, a small catheter (tube) containing the embryos is carefully inserted through the cervix and into the uterine cavity. This is guided with an ultrasound to ensure that they are gently placed in the proper location. After the transfer, the patient relaxes for approximately 15 minutes then is able to go home. Once placed in the uterus, the embryos cannot “fall” out or dislodge.
After the Embryo Transfer
After the retrieval and transfer, hormone supplementation (using progesterone) is continued to provide extra support for the pregnancy. Progesterone is available in several forms including injection, vaginal tablets, and vaginal creams. A blood pregnancy test is performed about 14-16 days after the egg retrieval. Once a pregnancy test is positive, an ultrasound is performed approximately 3 weeks later, at which time we should be able to detect a heartbeat in the embryo. Progesterone is continued for an additional 2-3 weeks after the detection of a heartbeat.
Summary
At first glance, the steps of IVF appear very complicated. However, our entire team of physicians, nurses, and staff will work to guide you step-by-step through the entire process. Having a dedicated team available and on call 24-hours a day ensures that you have a continuous support network at your fingertips. The entire time-frame from the start of a birth control pill until the pregnancy test is approximately 7 weeks, during which specific IVF medications are used for 4 weeks. IVF is not necessary for every couple, but it is always the fastest route to a pregnancy.
I previously discussed the impact that fibroids can have in a woman’s life, primarily that of irregular bleeding, pelvic pain, and infertility. There are various options for the treatment of fibroids depending on the future fertility plans of the woman. The guiding question should be whether or not there is or may be any future plans to conceive. If so, then the only treatment that should really be considered is a myomectomy, a surgical procedure in which the fibroids are removed, leaving the uterus in place. If childbearing is complete, then there are several other routes which may be chosen. These could include a myomectomy, hysterectomy, uterine artery embolization, endometrial ablation, or MRI guided focused ultrasound.
Medical (Non-Surgical) Management of Fibroids
I did not mention the use of medications in the above paragraph due to the fact that this is not a good long term option for fibroid treatment, and is only used in a few circumstances. The medications that are used work by shutting down the ovary temporarily. This induces a temporary reversible menopause-like situation. This can be used to reduce bleeding due to fibroids, and help to shrink them by about 30%. However, this effect only lasts as long as the medication is used, with fibroids returning to their normal size soon after stopping the medication. Due to the menopause-like situation, this treatment should only be used for short term treatment (<1 year duration) with ultimate definitive therapy. This is most frequently used in women who are having heavy bleeding, allowing the bleeding to slow or stop down so that her body can recover before ultimately having surgery.
Myomectomy
In a myomectomy, uterine fibroids are surgically removed, leaving the uterus in place. If fibroids are located primarily within the uterine cavity (submucosal type), then they may be able to be removed with a hysteroscope (small camera) by entering the uterus through the cervical opening. In most cases, they are removed by making an incision on the outside of the uterus, removing the fibroids, and suturing (sewing) the uterus back together. In the past, myomectomies were performed through large open incisions in the abdomen, often going side-to-side, but sometimes vertically. This type of “open” myomectomy has significant drawbacks, including a 2-3 day hospital stay, extended recovery, adhesion (scar tissue) formation, and significant blood loss. In recent years, there has been a large shift towards performing myomectomies by laparoscopy (through small incisions using cameras). This “minimally invasive” route offers significant advantages, including less pain, a more cosmetic result with smaller incisions, usually no overnight hospitalization, less bleeding and scar formation, and a much faster recovery. Before a few years ago, only some selected cases were performed laparoscopically due to the significant increase in difficulty in performing myomectomies laparoscopically. The introduction of the da Vinci Robotic System for laparoscopic assistance has made a tremendous improvement, allowing nearly any myomectomy to be performed laparoscopically, regardless of size or difficulty. Our practice specializes in myomectomies and is able to repair and reconstruct the most severely distorted uterus due to fibroids.
Hysterectomy
For some women, they may choose to have a hysterectomy (removal of the uterus) as a treatment for fibroids. It is for this reason that a hysterectomy has become one of the most common surgeries that a woman may face during her lifetime. Most hysterectomies can be accomplished by a minimally invasive route, either removing it through the vagina, or laparoscopically through the use of small cameras, or in difficult cases, using Robotic assistance to perform it through a camera. Rarely is an open incision needed for a hysterectomy.
Uterine Artery Embolization
A uterine artery embolization (UAE) procedure performed by a specially trained radiologist which involves injecting small particles (about the size of a small grain of sand) into the uterine blood vessels. These particles clog the small blood vessels that supply the fibroids, cutting down the blood supply and causing the fibroids to die and degenerate. Fibroid volume shrinks by 40% to 50%, and the majority of patients experience symptomatic relief. However, patients generally experience several days of pain after the procedure and are usually hospitalized for 1-2 days. Women who are older may at risk of developing early ovarian failure. Due to the effect on uterine blood flow, this technique is not recommended for women who would like to conceive in the future, and for those who do, there is a slightly higher rate of pregnancy complications.
Endometrial Ablation
An endometrial ablation is a procedure that is intended to treat heavy uterine bleeding in general. This could be due to fibroids or to other causes. In this procedure, one of several techniques are used to basically burn or cut out the endometrium (lining of the uterus). This significantly limits the amount of bleeding that occurs. An ablation may be effective in treating bleeding due to fibroids, but may often provide only effective treatment for a limited time (perhaps 1-3 years). Bleeding may return as fibroids continue to grow and enlarge.
MRI Guided focused ultrasound
This is a newer approved technique which uses powerful ultrasound waves to destroy the fibroid. It has only limited availability, and only small studies available with long term follow up. It is likely most effective for someone with a small number of larger fibroids. However, due to the lack of long term follow-up results it is not widely recommended and cannot be recommended for anyone wishing to retain their fertility.
In summary, if fertility is still desired, then a myomectomy is the procedure of choice. Other forms of fibroid treatment should not be undertaken due to known problems with subsequent pregnancies. If childbearing is not desired, then the woman can make an informed decision with her physician regarding her desires, knowing that each of the above option has both benefits and drawbacks.
Uterine fibroids, also called myomas or leiomyomas, are benign (non-cancerous) tumors that arise from the muscle of the uterus. They are extremely common, with most women developing them at some point in time during their life. One study found that by the age of 50, that 70% of Caucasians and 80% of African-Americans will have developed fibroids. There appears to be a genetic link to the development of fibroids, often arising earlier and growing faster in African-Americans. Fibroids often appear to begin when a woman is in her 20s, and usually grow at a slow rate, only reaching a size large enough to cause symptoms when she is in her 30s-40s. Because of this, problematic fibroids are the most common indication for a hysterectomy in women.
When they reach a large enough size, they can cause symptoms such as heavy or irregular bleeding, infertility, pain, or symptoms of “pressure” by pressing on other pelvic organs. However, many fibroids can be totally asymptomatic, with the woman not having any problems until they are incidentally seen by an ultrasound or CT scan. 
We classify the location of fibroids as primarily four different types: 1) subserosal- located in the outer-most portion of the wall of the uterus, just underneath the outer surface (the serosa); 2) pedunculated- a type of subserosal fibroid that lies mostly outside of the uterus and only connected by a small stalk of tissue; 3) intramural- found within the main body of uterus; 4) submucosal- In the inner-most portion of the uterus and protruding into the cavity of the uterus. About 55% of fibroids are subserosal or pedunculated; 40% are intramural; and 5% are submucosal. As fibroids increase in size, they may come to span two or more of these categories. Typically, submucosal and larger intramural fibroids are responsible for heavy menstrual bleeding.
So do they affect your chance of conceiving? In short, likely yes, possibly no. This depends on the location and size of fibroids. Many studies have tried to determine the right answers to this question, with some progress being made. Some of the results are still somewhat debated. Many women with fibroids conceive on their own and they may only be discovered during routine pregnancy ultrasounds. However, for many women they can play a role in hindering fertility. It is estimated that fibroids play a role in approximately 5-10% of infertility cases.

There is very good data and it is clear that submucosal fibroids can decrease pregnancy rates by about 35%, and increase miscarriage rates by over 65%. On the other hand, fibroids that are only of the pedunculated or subserosal type appear to have no effect on fertility or miscarriage. Fibroids that are intramural do appear to decrease fertility though the scientific data is less clear. With all categories, however, there likely is an effect of fibroid size and fertility impact. Subserosal fibroids can enlarge to the point that they become predominantly intramural or even submucosal. Once this size, they may also impact fertility.
Both the lowered pregnancy rate and increased miscarriage rate is likely due to altered blood flow within the uterus. The fibroids may serve to divert necessary blood flow away from the uterine cavity where an embryo could implant.
We’ll discuss the treatment options for fibroids in an upcoming blog topic.
I had a large cyst on my ovary, and was placed on a birth control pill to make it go away. Another doctor told me that the pill doesn’t make them go away any faster.
The doctor is correct, that cysts don’t go away any faster while on birth control pills than if not on them at all. However, many physicians use them when a cyst is found. The reasoning for this is that while on the pill, the brain’s normal hormone production that stimulates the ovary gets placed on hold. This prevents the brain from trying to start an ovulation on top of the already present cysts. If this occurred, the new follicles could then be later confused with the previous cysts on ultrasound, thus complicating the picture. Also, when on a birth control pill, once that the cyst has resolved, the physician can then just stop the pill at that point and immediately start ovulation medication if needed. If the patient is not on a pill, then they may have to wait a few extra weeks until their normal menstrual cycle begins in order to start ovulation treatment. So ultimately, in patients trying to conceive, the use of a birth control pill in this setting will always make the overall process shorter.
I had an HSG (x-ray dye study of the tubes) performed, and they didn’t see one tube. Does that mean I don’t have a tube? Why wasn’t I told this before?
Wait just a minute before panicking. I need to help explain some things. When you had the HSG performed, if the radiologist or doctor didn’t see your one fallopian tube, that does not necessarily mean that it is absent or not working at all. If a uterus is normal, then it would be extraordinarily rare for one or both tubes to be absent. When one tube doesn’t show up on the HSG, it is usually has a simple explanation. There is a small muscle at the opening (the ostia) that leads from the uterus into the tube. This is a round sphincter muscle, just like the anal sphincter. When dye is injected into the uterus, these muscles often spasm, closing down tightly. Once they do this, they do not relax easily. So if the dye goes out one tube, and the other tubal openins is having a spasm, the dye just follows the path of least resistance and continues to go out the open tube. With this happening, pressure cannot be built up inside of the uterus to overcome the spasm of the other tube opening.
If the fallopian tube filled with dye and was blocked at the end, then that is a different story. But of the tubes that don’t even fill up, well over 90% of them are normal. The tubal spasm is a side effect of the HSG and is sometimes more commonly found if the HSG is not carefully performed. There can be scar tissue at the opening causing it to be scarred shut, but that is not commonly seen. Tubal scar tissue usually affects the end of the tube and only rarely at the point where the tube joins the uterus. Occasionally there can be a polyp, or endometrial overgrowth, that is right in front of this opening and could serve to block the ostia. We used to take all of these patients to laparoscopy to investigate this, and after doing this time and time again, what we realized was that nearly all of them were normal at the time of surgery. Going to surgery is still a reasonable option for some patients, though, depending on the situation. If one tube appears to be working normally, then more conservative therapy could be used.










